Student Guide Sections
Introduction to Chemistry and Energy Efficiency
Insulation
Fertilizer and Crop Production
Lighting
Polymers
Introduction to Chemistry and Energy Efficiency
What role does chemistry play in our lives? Chemistry improves our standard of living. If we take a snapshot of any moment in our lives, chemistry’s effects can be measured. The chemistry industry has made our lives more productive and sustainable by providing us with the convenience of synthetic materials such as plastic, foam polystyrene and fabric; advancing our quality of living with pharmaceuticals, cosmetics, and cleaning products; and improving our ability to produce food and purify water.
The population of the Earth is increasing while its resources are not, creating potential imbalances in supply and demand. The chemistry industry has introduced products and perfected technologies that will meet the needs of society and help to sustain our resources for an ever-growing population. The goods produced are designed to increase safety, production and efficiency while simultaneously minimizing the impact on resources and the environment.
Climate Change
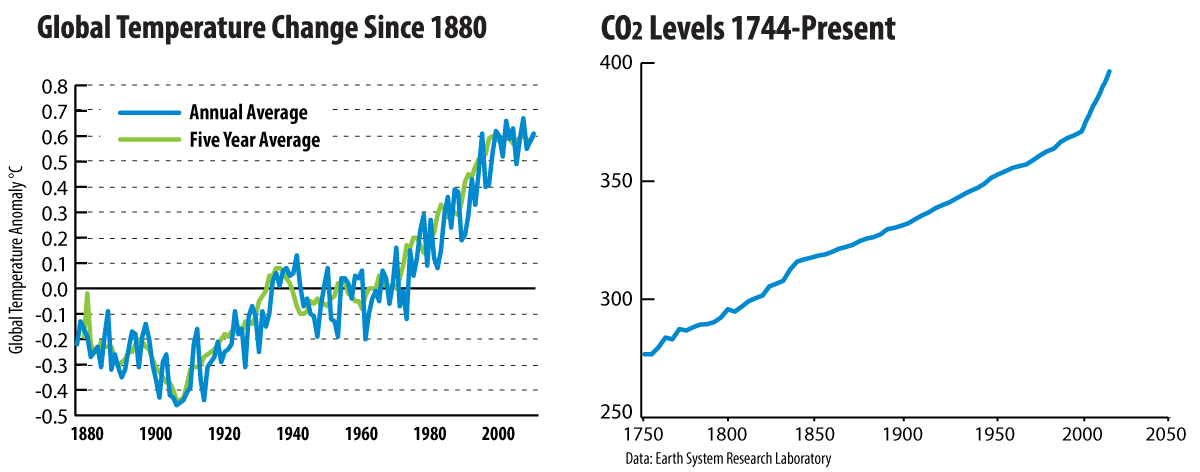
People demand products to support their lifestyles. This demand has a direct impact on our environment. Working over the last several decades, scientists have shown that for the first time ever, human activities are altering the climate; the data supporting this conclusion continues to grow in depth and breadth. Since the Industrial Revolution began, research shows that the concentrations of greenhouse gases (GHGs), which trap heat in the atmosphere, have increased. The most prevalent greenhouse gas studied is carbon dioxide (CO2), which is produced from burning fossil fuels such as coal and petroleum. A rise in global temperatures corresponds to the rise in carbon dioxide levels in the atmosphere.
Energy from the sun passes through the atmosphere in the form of electromagnetic radiation, much of which reaches the Earth’s surface. The atmosphere, made of a mixture of gases including water vapor, absorbs some of the solar radiation. At the earth’s surface, the radiant energy from the sun is transformed into thermal energy. Most of the energy is reflected from the surface of the Earth back towards space. When greenhouse gas concentrations are at normal levels, most thermal energy continues out into space. However, when greenhouse gas concentrations are high, the thermal energy is absorbed by the greenhouse gases, heating the atmosphere. The reason that much of this energy remains in the atmosphere is due to the properties of greenhouse gases.
The atmosphere is composed mostly of nitrogen and oxygen. The rigid bonding structure of nitrogen (triple bonds) and oxygen (double bonds) do not allow for much flexibility with the bending, vibration, and stretching of their bonds. The greenhouse gases of carbon dioxide, methane, water, and nitrous oxide have a great deal of flexibility in the bonds of each molecule. Therefore, as the molecules are exposed to radiant energy, the bonds within these molecules have a great deal of potential to absorb it and transform it to thermal energy.
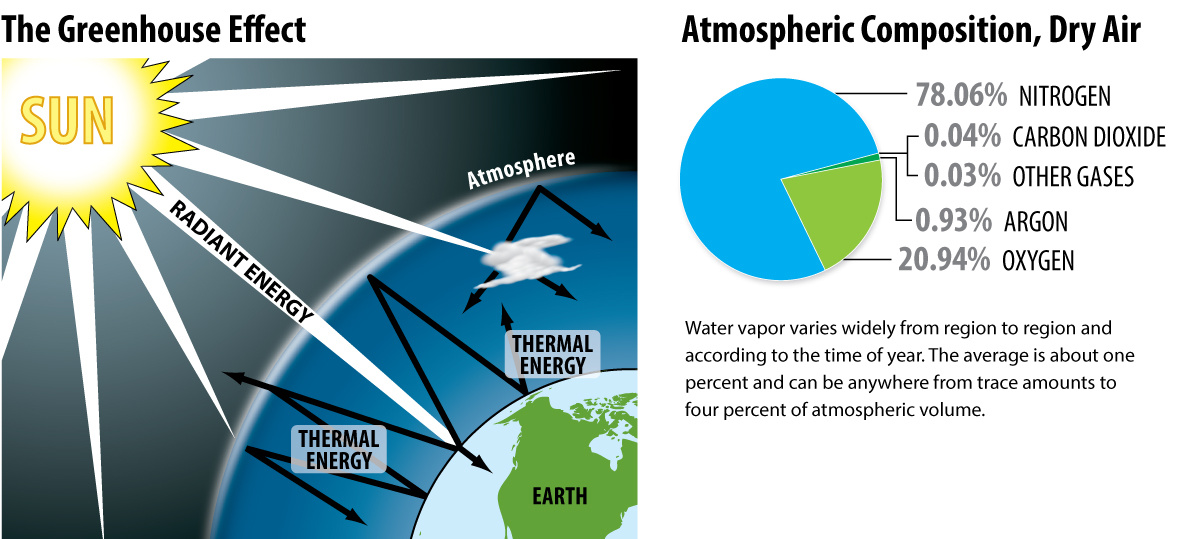
It is the increase of the kinetic energy within the bonds of these molecules that raises the internal energy of these molecules and can then be transmitted to other atmospheric gases to increase the temperature of the atmosphere. The result is a net increase in the average kinetic energy of the atmosphere gases, and an increase in the temperature of the land and water on the Earth’s surface.

Chemistry and Climate Change
As concern for the impact of human activity on global climate patterns becomes more prominent in the world, many people are working to change behaviors to reverse human impact on the environment. The chemistry industry is committed to not only reducing the amount of greenhouse gas emissions from the production process, but also producing products that use less energy and draw on renewable energy sources.
The International Council of Chemical Associations (ICCA) commissioned a report to analyze the impact chemical products have on climate change. The report, called The McKinsey Report, was released in 2009. The analysis showed that by applying new technology to reducing greenhouse gas emissions, substantial decreases in the amount of greenhouse gases produced by other industries and consumers can be achieved. For one unit of greenhouse gases emitted by the chemistry industry, 2-3 fewer units of greenhouse gases were subsequently produced when using the new technology. According to data cited within the report, the chemistry industry has increased its production world-wide over the last 20-30 years, while at the same time reducing its total energy consumption. The study looked at more than 100 products and their role in reducing greenhouse gases during their life cycles. The most significant emissions savings by volume came from building insulation materials, agrochemicals, and lighting improvements.
Nutrient Cycles
To gain a clear understanding of the role the chemistry industry has in reducing greenhouse gases, it is necessary to look at the role two chemical elements have in ecosystems. The first, carbon, is exchanged among different reservoirs (atmosphere, biosphere, lithosphere, and hydrosphere) in a biogeochemical exchange process known as the carbon cycle. Carbon is found in both living, or biotic, compounds, as well as non-living, or abiotic, compounds. Examples of biotic compounds are proteins and carbohydrates. Abiotic carbon compounds include carbonates in limestone and carbon dioxide in the atmosphere. The movement of carbon among the earth’s reservoirs – the atmosphere, biosphere, lithosphere, and hydrosphere – can be a very slow process. The ocean and lithosphere historically have been large sinks for the storage of carbon. Scientists are most concerned with the way human activity has shifted the distribution of carbon among the various reservoirs, especially toward greater concentrations in the atmosphere.
Watch a video about the carbon cycle.
Another biogeochemical cycle is the exchange of nitrogen among the biosphere, atmosphere, lithosphere, and hydrosphere, known as the nitrogen cycle. Elemental nitrogen (N2) exists as the most prevalent gas in the atmosphere. Nitrogen is biologically inactive and therefore harmless. However, nitrogen atoms are extremely important in the structures of proteins and nucleic acids in all living things. Nitrogen is also found in abiotic compounds containing nitrates and ammonia within the lithosphere and hydrosphere. Because most organisms cannot use atmospheric nitrogen, it must be chemically reacted into a compound that can be used. Bacteria within the soil are able to reduce nitrogen and produce ammonia in a process called nitrogen fixation. This natural process is one way that nitrogen has moved from the atmosphere to the soil which is part of the biosphere reservoir. Human activity has altered the nitrogen cycle by creating an additional mechanism by which nitrogen can be fixed into biologically active compounds. The chemical production of nitrates and ammonia from nitrogen gas has increased as the demand for food supplies has also increased. Nitrates and ammonia are both used as components of fertilizers.
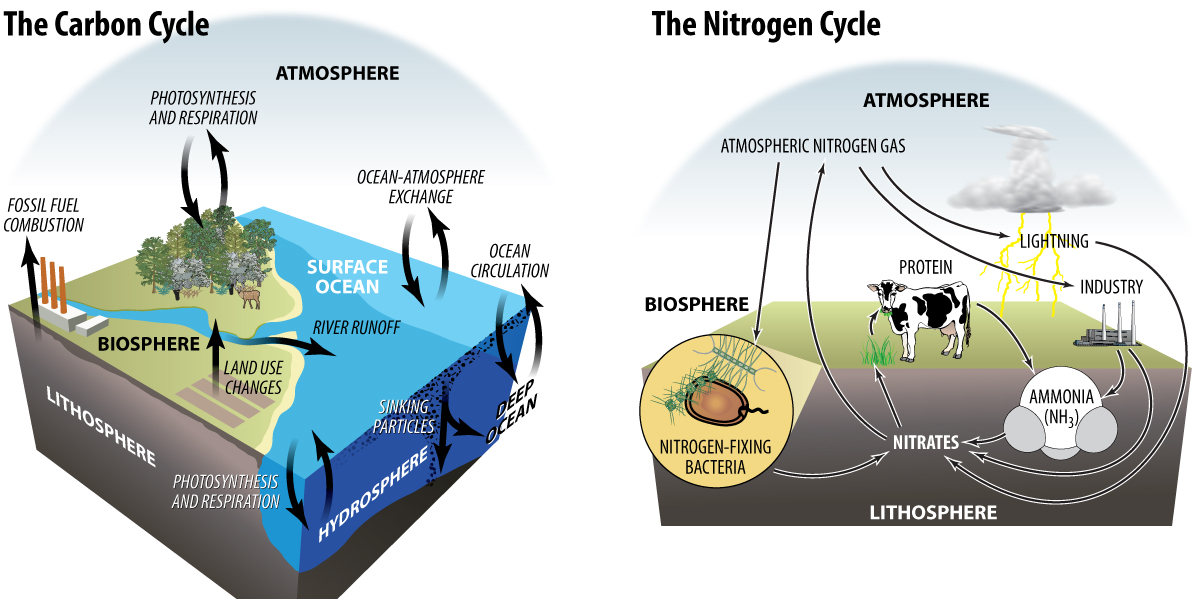
The cycling of nutrients through ecosystems has been a natural process since the beginning of time. The transfer of the different elements through the various reservoirs has been a process that can take a few years to several hundred years. Yet, as human activities have increased, the rate of exchange of elements among reservoirs has escalated. Human activity has shifted the concentration these elements to an imbalanced level within reservoirs. As society’s demand for products that improve and support lifestyles continues to increase, there are trade-offs. We must consider how to best meet societal needs while minimizing negative impacts these products and the processes that produce them have on nutrient cycles and the environment overall.